 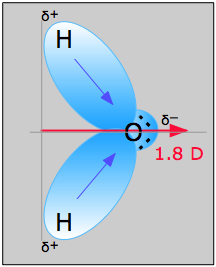
The PH3 molecule has one lone pair and three bond pairs. 
Here is the increasing order of repulsion forces:īond pair – bond pair < bond pair – lone pair < lone pair – lone pair This force is lower between a lone pair and a bond pair, whereas it is the lowest between two bond pairs of electrons. The number of lone pairs and bonds affect the shape of a molecule.Įvery kind of electron pair repulse the other pairs the force of repulsion is maximum between the two lone pairs. It reduces the repulsion between the valence electrons, thus helping the molecule get a stable structure. If you have studied the VSEPR theory, you know that each pair of electrons tend to stay at the maximum possible distance from one another. The shape of a molecule is defined by how many lone pairs and the number of covalent bonds it has. The two remaining electrons form a lone pair. The molecular geometry of a compound is determined by two factors the Lewis structure and the VSEPR (valence shell electron pair repulsion) theory.įrom the Lewis molecular structure of PH3, we have seen the phosphorous atom has five valence electrons.ĭuring the bonding process, Phosphorous is surrounded by three hydrogen atoms, and each is connected by a single bond. In the following section, we will learn in detail about the geometric shape of this compound. The geometric structure of this compound is a trigonal pyramid in shape. So, the combination has a stable structure now. Now, if you check the surrounding electrons of both the compounds, you can see each Hydrogen atom has two surrounding atoms, while the phosphorous atom has eight electrons around it. The remaining two unpaired electrons of phosphorous are placed on the 4th side that forms a lone pair. Three valence electrons of phosphorous forms pairs with three valence electrons from the hydrogen atoms. Since hydrogen has one valence electron, and phosphorous has three, so P is the central atom in the molecular structure of this compound. The electronic configuration of the atoms let us know how many atoms can participate in the bonding. In Phosphene, three hydrogen atoms combine with phosphorous. It means hydrogen has one valence electron while phosphorous has three. If you look at the periodic table, you will see hydrogen is placed in the first column while phosphorous in the 5th column. Phosphorous has an electronic configuration of 1S2 2S2 2P6 3S2 3P3. Hydrogen has an electronic configuration of 1S1 as it has only one electron. To understand the structure of PH3, we should know the electronic configuration of the atoms and how many valence electrons are there in the atoms. Lone pair electronsĪffect both the partial charges and the distance between partial charges.The molecular formula of phosphene is PH3 which indicates the compound has one phosphorous atom bonding with three hydrogen atoms. Molecule and the distance in which they are separated. Aĭipole moment is the product of the magnitude of the partial charges on the 
Pair electrons have effect on the net dipole moment of a molecule. Resulting in a low molecular dipole moment. That the dipole due to the lone pair opposes the N-F bond dipoles, Of the N-F bonds is opposite to that of the N-H bonds in ammonia, so Fluorine is more electronegative than nitrogen and the polarity In contrast to NH 3,NF3 has a much lower dipole moment of 0.24 D. There isĪlso a dipole associated with the lone pair and this reinforces theĬontribution made by the polar covalent N-H bonds to ammonia's dipole moment. That the N-H bonds are polar with a net negative charge on the nitrogenĪtom and a smaller net positive charge on the hydrogen atoms. Of nitrogen (3.04) is greater than that of hydrogen (2.2) the result is Lone pairs can make a contribution to a molecule's dipole moment. Lone pair is a valence e- pair which is not shared with another atom and is sometimes called a non-bonding pair.For ex. Concepts of Physics by HC Verma for JEE.IIT JEE Coaching For Foundation Classes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
